Project Description
For this project, we had to design a board game with rules, instructions, an overarching theme, and game pieces. On top of that, we also had to incorporate different types of chemical reactions into our board game. Each board game was to include a single displacement reaction, double displacement reaction, production of a gas, lighting of an LED, chemical change, and a physical change. Along with our board game, we needed to create a detailed instructions sheet, safety requirements sheet, detailed explanations of all our chemical reactions, an intricate blueprint of our board game, and game pieces. We worked on our project for approximately three to four weeks, and at the end of our work period, we had to present our board games at the "Family Game Night" where parents came to school one evening and played and reviewed our game. For my groups board game our theme was Christmas Candyland.
Our Project
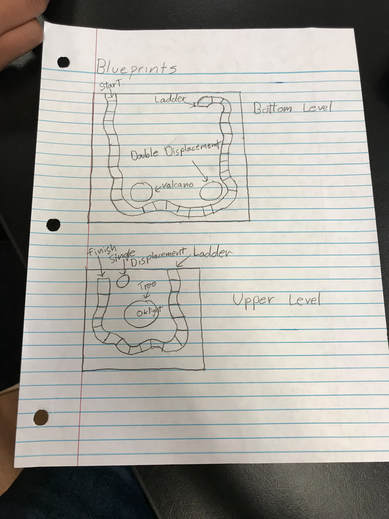
Our board game was based around the classic game Candyland except with a Christmas twist. Each player gets a game piece and whenever it's their turn draws a card and move to that color on the board which corresponds to the card color. Whenever you land on a special square you get to do a chemical reaction. This is where our single displacement, double displacement, gas production, and LED lighting reactions occurred. You must complete the reaction correctly to advance. If you fail to do so you can't move on until you completed successfully. Our chemistry aspect of this project was quite fun and informative. For our single displacement reaction we added a blue solution known as copper nitrate to aluminum foil. The aluminum was connected to a breadboard with an LED, and when the copper nitrate dissolved the aluminum, the electrical circuit would be completed and the LED would light. For our double displacement reaction we combined potassium iodide with lead nitrate. Combining these two results in a change of color going from two clear liquids to one bright yellow liquid. Upon being combined the two chemicals react with each other combining them to be potassium nitrate and lead iodide. Because they react with the opposite component it is considered a double displacement reaction. For our production of a gas we made a Christmas volcano and combined baking soda and vinegar. To win the game one must land on the final special square and light the LED on the top of the Christmas tree.
Below are the pictures of our game board.
Below you can find our game instructions, chemical equations, and safety guidelines!
Major Concepts
-Single Displacement Reaction: (A+BC = B+AC) an element reacts with a compound and takes the place of another element in that compound. Our single displacement reaction was our aluminum and copper nitrate reaction.
-Double Displacement Reaction: (AB+CD = CB+AD) two compounds react and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products. Our double displacement reaction was the combining of potassium iodide and lead nitrate to form a yellow solution.
-Synthesis Reaction: (A+B = AB) multiple reactants combine to form a single product and release energy in the form of heat and light. No synthesis reactions were used in our project.
-Decomposition Reaction: (AB = A+B) single compound breaks down into two or more elements or new compounds and usually involves an energy source. No synthesis reactions were used in our project.
-Polyatomic Ions: a charged ion composed of 2 or more atoms covalently bonded to act as single unit
-Cation: positively-charged ion (tend to be metals)
-Anion: negatively-charged ion (tend to be nonmetals)
-Reactant- When chemical reactions are written down, the reactants are written on the left side. These react to form products. Reactants were crucial in our project, such as copper nitrate and aluminum for single replacement, and potassium and lead for our double displacement. These cause the reaction to occur.
-Product- form from reactants. Products are a substance that is formed as the result of a chemical reaction always on the right side of a chemical equation. Many products were created by performing chemical reactions such as the reaction from the baking soda and vinegar and the potassium iodide and lead nitrate creating potassium nitrate.
-Double Displacement Reaction: (AB+CD = CB+AD) two compounds react and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products. Our double displacement reaction was the combining of potassium iodide and lead nitrate to form a yellow solution.
-Synthesis Reaction: (A+B = AB) multiple reactants combine to form a single product and release energy in the form of heat and light. No synthesis reactions were used in our project.
-Decomposition Reaction: (AB = A+B) single compound breaks down into two or more elements or new compounds and usually involves an energy source. No synthesis reactions were used in our project.
-Polyatomic Ions: a charged ion composed of 2 or more atoms covalently bonded to act as single unit
-Cation: positively-charged ion (tend to be metals)
-Anion: negatively-charged ion (tend to be nonmetals)
-Reactant- When chemical reactions are written down, the reactants are written on the left side. These react to form products. Reactants were crucial in our project, such as copper nitrate and aluminum for single replacement, and potassium and lead for our double displacement. These cause the reaction to occur.
-Product- form from reactants. Products are a substance that is formed as the result of a chemical reaction always on the right side of a chemical equation. Many products were created by performing chemical reactions such as the reaction from the baking soda and vinegar and the potassium iodide and lead nitrate creating potassium nitrate.
Our Original Blueprint
Reflection
This project was by far one of the most fun projects I have done in all my years in STEM. For the most part this project went well but it had its ups and downs. My group wasn't exactly the most studious, but we got the job done. We definitely had a lot of fun during this project because it was really interesting to learn about reactions we can do in chemistry that are easy and fun to do. We all had a job throughout the process of the project and we all completed it well. Our group collaboration was definitely one of our stronger points for both projects. We each took a task in the project and sought it out to the end. Each person did their part well and that definitely contributed to our success. I think something that could have gone better would be our planning strategy. We had an idea of what we wanted to do but at times struggled to execute that idea flawlessly. In this project I really listened to my group members opinions and didn't just do what I wanted. Working as a team is really beneficial toward the end goal for the project. Although my leadership and cooperation weren´t excellent in this project, I did pick up the work in areas we were lacking. One of our group members was not here for a big chunk of the time so the rest of the group really had to work a lot harder in order to be ready for our presentation night. Two things I can improve on for future projects could be being more empathetic and more open to new ideas. BY making this change my group can get more work done and have our presentation be the best it can be.